Cooling Curves And Its Types Construction Of Phase Diagrams
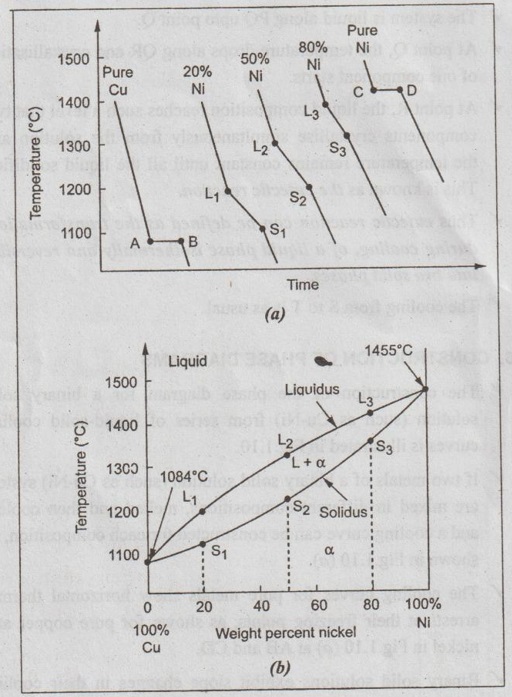
Cooling Curves And Its Types Construction Of Phase Diagrams By removing the time axis from the curves and replacing it with composition, the cooling curves indicate the temperatures of the solidus and liquidus for a given composition. this allows the solidus and liquidus to be plotted to produce the phase diagram: this page titled 12.5: interpretation of cooling curves is shared under a cc by nc sa. The cooling curve method is one of the oldest and simplest methods to determine phase diagrams and phase transition temperatures. this is achieved by recording temperature (t) of a material versus time as it cools from its molten state through solidification (at constant pressure). whenever a phase change takes place in a metal or alloy, the.

Practical Maintenance в Blog Archive в Phase Diagrams Part 1 One component phase diagram. figure 1 illustrates the temperatures and pressures at which water can exist as a solid, liquid or vapor. the curves represent the points at which two of the phases coexist in equilibrium. at the point tt vapor, liquid and solid coexist in equilibrium. in the fields of the diagram (phase fields) only one phase exists. A cooling curve for a sample that begins at the temperature and composition given by point a is shown in figure 8.10.1b 8.10. 1 b. figure 8.10.1 8.10. 1: (a) cooling of a two component system from liquid to solid. (b) cooresponding cooling curve for this process. as the sample cools from point a, the temperature will decrease at a rate. Phase diagram and “degrees of freedom”. phase diagrams is a type of graph used to show the equilibrium conditions between the thermodynamically distinct phases; or to show what phases are present in the material system at various t, p, and compositions. “equilibrium” is important: phase diagrams are determined by using slow cooling. Interpretation of cooling curves. the melting temperature of any pure material (a one component system) at constant pressure is a single unique temperature. the liquid and solid phases exist together in equilibrium only at this temperature. when cooled, the temperature of the molten material will steadily decrease until the melting point is.

Heating And Cooling Curves вђ Overview Examples Expii Phase diagram and “degrees of freedom”. phase diagrams is a type of graph used to show the equilibrium conditions between the thermodynamically distinct phases; or to show what phases are present in the material system at various t, p, and compositions. “equilibrium” is important: phase diagrams are determined by using slow cooling. Interpretation of cooling curves. the melting temperature of any pure material (a one component system) at constant pressure is a single unique temperature. the liquid and solid phases exist together in equilibrium only at this temperature. when cooled, the temperature of the molten material will steadily decrease until the melting point is. 13.2.2 solid–liquid systems. figure 13.1 temperature–composition phase diagram for a binary system exhibiting a eutectic point. figure 13.1 is a temperature–composition phase diagram at a fixed pressure. the composition variable is the mole fraction of component b in the system as a whole. The most common approaches for the determination of phase diagram of two component systems are “cooling curve” and “thaw melt” methods. these methods are quite popular due to their easiness and practicability to many systems. 1. cooling curve method: in this approach, a liquid mixture of two components a and b at temperature t is.

Comments are closed.